4-AMINOMETHYLPHENYLBORONIC ACID, HYDROCH
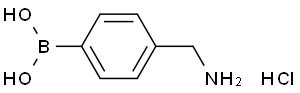
4-Aminomethylphenylboronic acid hydrochloride
CAS: 75705-21-4
Molecular Formula: C7H11BClNO2
4-AMINOMETHYLPHENYLBORONIC ACID, HYDROCH - Names and Identifiers
| Name | 4-Aminomethylphenylboronic acid hydrochloride |
| Synonyms | TIMTEC-BB SBB003689 4-Boronobenzylamine, HCl 4-AMINOMETHYLPHENYLBORONIC ACID HCL [4-(aminomethyl)phenyl]boronic acid 4-AMINOMETHYLPHENYLBORONIC ACID, HYDROCH 4-AMINOMETHYLPHENYLBORONIC ACID HYDROCHLORIDE 4-Aminomethylphenylboronic acid hydrochloride 4-AMINOMETHYLBENZENEBORONIC ACID HYDROCHLORIDE 4-Aminomethylphenylboronic acid, hydrogen chloride |
| CAS | 75705-21-4 |
| EINECS | 672-309-4 |
| InChI | InChI=1/C7H10BNO2/c9-5-6-1-3-7(4-2-6)8(10)11/h1-4,10-11H,5,9H2 |
4-AMINOMETHYLPHENYLBORONIC ACID, HYDROCH - Physico-chemical Properties
| Molecular Formula | C7H11BClNO2 |
| Molar Mass | 187.43 |
| Density | 1.18g/cm3 |
| Melting Point | 240-250 |
| Boling Point | 337.7°C at 760 mmHg |
| Flash Point | 158.1°C |
| Vapor Presure | 4.02E-05mmHg at 25°C |
| Appearance | Powder |
| Color | Pale yellow |
| Storage Condition | Keep in dark place,Inert atmosphere,2-8°C |
| Refractive Index | 1.567 |
4-AMINOMETHYLPHENYLBORONIC ACID, HYDROCH - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R22 - Harmful if swallowed |
| Safety Description | S37/39 - Wear suitable gloves and eye/face protection S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| HS Code | 29310095 |
| Hazard Class | IRRITANT |
4-AMINOMETHYLPHENYLBORONIC ACID, HYDROCH - Reference Information
| introduction | 4-aminomethylphenylboronic acid hydrochloride is a 4-aminomethylphenylboronic acid compound. 4-Aminomethylphenylboronic acid compounds are one of the substrates commonly used in Suzuki reactions. They are often used in the synthesis of biphenyl compounds and are widely used in the fields of pharmacy and OLED materials. There are reports that it can be used to prepare BTK inhibitors. |
| use | 4-aminomethylphenylboronic acid hydrochloride is an organic compound of carboxylate and can be used as a pharmaceutical intermediate. |
| preparation | the first step: add 160mL of water, 80mL of methanol, 10g of tert-butyl carbamate (85.36mmol), 30.4g of sodium p-toluenesulfonate (170.72mmol), 12.8g 4-formyl phenylboronic acid (85.36mmol) and 6.5mL of formic acid to the reaction bottle in sequence, after 36h of reaction at room temperature, the crude product was obtained by suction filtration, washed with water, washed with methyl tert-butyl ether, and dried to obtain a white solid. Then, white solid and 600mLTHF are added into the reaction bottle, 99.5g of potassium carbonate (720mmol) are added into the reaction bottle, heated to 60 ℃ for 15h, cooled, filtered by diatomite, concentrated and evaporated to obtain 15.14g of imine intermediate with a yield of 71.2%; Step 2: Dissolve 15.14g of imine intermediate (60.78mmol) in 150mL of methanol, add 4.6 gNaBH4(121.56mmol) in batches, and react overnight at room temperature, concentrate under reduced pressure to obtain a reduction intermediate. The reduction intermediate was adjusted to pH = 5-6 with 1.0M citric acid aqueous solution, extracted with ethyl acetate, and the organic phase was washed and dried to obtain 4-(N-Boc-aminomethyl) phenylboronic acid. Step 3: Add 4-(N-Boc-aminomethyl) phenylboronic acid to the reaction flask, then add 100mL 3.0M hydrogen chloride/ethyl acetate solution (), stir at room temperature for 2 hours, and filter to obtain 10.18g of 4-aminomethyl phenylboronic acid hydrochloride with 89.4% yield. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 4-Aminomethylphenylboronic acid hydrochloride Visit Supplier Webpage Request for quotationCAS: 75705-21-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-Aminomethylphenylboronic acid hydrochloride Request for quotation
CAS: 75705-21-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 75705-21-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Multiple Specifications
Product Name: 4-AMINOMETHYLPHENYLBORONIC ACID HYDROCHLORIDE Request for quotationCAS: 75705-21-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 4-Aminomethylphenylboronic Acid Hydrochloride Visit Supplier Webpage Request for quotation
CAS: 75705-21-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 75705-21-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 4-Aminomethylphenylboronic acid hydrochloride Visit Supplier Webpage Request for quotationCAS: 75705-21-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4-Aminomethylphenylboronic acid hydrochloride Request for quotation
CAS: 75705-21-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 75705-21-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Multiple Specifications
Product Name: 4-AMINOMETHYLPHENYLBORONIC ACID HYDROCHLORIDE Request for quotationCAS: 75705-21-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 4-Aminomethylphenylboronic Acid Hydrochloride Visit Supplier Webpage Request for quotation
CAS: 75705-21-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 75705-21-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



